Abstract
Persisting dizziness (i.e., remaining disabling for several months post-injury) is a common physical symptom of mild Traumatic Brain Injury (mTBI) that influences the performance of activities of daily living. Yet, the mechanism of post-injury dysfunction is not fully understood, as it is often assessed and treated in conjunction with other symptoms. In order to garner a better understanding of dizziness in mTBI and effective treatment for mTBI-related dizziness, subjective reports before and after a therapy, treatment, or intervention must be compared. The current research used the Dizziness Handicap Inventory (DHI) to assess dizziness in clients (N = 75) with mTBI before and after a daily intensive treatment program with a short duration (i.e., 2-4 weeks) at a physiotherapy clinic in Southwestern Ontario, which included soft tissue release, specialized electrotherapy (Myoworx TM20 device), and exercise therapy. Statistically significant effects after treatment only emerged in physical subscales, where female clients had greater scores. Closer inspection of confidence intervals revealed clinically meaningful differences in total DHI scores, functional, and emotional subscales. As such, findings offer additional support for the use of soft tissue release, specialized electrotherapy, and exercise in the treatment of dizziness related to mTBI.
Keywords: Traumatic Brain Injury, Concussion, Dizziness, Treatment
The World Health Organization has estimated that mild traumatic brain injury affects more than 6/1000 people a year (Cassidy et al., 2004). In Canada, a surveillance study of electronic medical records revealed there were 15,086 cases of concussion among a population of 1,052,141 primary care patients from 2010 to 2016. An increase in concussion rates was reported during this time period, with higher prevalence rates in males (1.48% vs. 1.35%) and among those ages 10-14 and 15-19 (Bang et al., 2019).
Mild traumatic brain injury (mTBI) and concussion are defined by the Ontario Neurotrauma Foundation (2018) as “the acute neurophysiological effects of blunt impact or other mechanical energy applied to the head, neck or body with transmitting forces to the brain” (p. 1). Common causes include sports injuries, motor vehicle collisions, bicycle accidents, falls, and assaults (e.g., Cassidy et al., 2004). Although the terms “concussion” and “mTBI” are often used interchangeably in the literature, it is important to note that concussions are a subset of mTBI and the term concussion is more commonly used in sports. In such instances of intracranial injury or persistent neurological impairment, mTBI is differentiated from concussion. (McCroy et al., 2017; Ontario Neurotrauma Foundation, 2018). We will henceforth use the term mTBI.
Defining diagnostic features of mTBI include balance impairment, behavioural change, cognitive dysfunction, and may also include headache, amnesia, and neurologic deficits (McCrory et al., 2017). Long-term outcomes include reduced functional ability, emotional distress, and delayed return to work or school (Ontario Neurotrauma Foundation, 2014). Overall, outcomes are generally favourable (Naalt, 2001), as most individuals tend to recover in 48 to 72 hours, and with the symptoms and signs usually resolve within 10 days (Halstead & Walter, 2010). However, in 15-30% of cases, post-concussive symptoms may persist for weeks or months and thus results in functional disability and an overall decline in quality of life (Barlow, 2016). Many factors contribute to symptom persistence (e.g., injury severity, previous TBI, etc.; Barlow, 2016), which may indicate post-concussion syndrome (PCS).
Persisting dizziness (i.e., remaining disabling for several months post-injury) is a common physical symptom of mTBI (Chamelian & Feinstein, 2004; Fife & Kalra, 2015; Hoffer et al., 2007; Ontario Neurotrauma Foundation, 2014; Willer & Leddy, 2006) which may impact activities of daily life (Maskell et al., 2007; Campbell & Parry, 2005). It is estimated that 23.8% to 81% of patients with mTBI may be affected (Hartvigsen et al., 2014; Maskell et al., 2006). In a study of 1716 patients with mTBI, dizziness was reported in 38.9% of patients 6 weeks post-injury, and 25.4% 1 year after injury (Hartvigsen et al., 2014). The incidence of persisting dizziness tends to be associated with injury severity and time since injury (Chamelian & Feinstein, 2004; Maskell et al., 2006). Patient descriptions of dizziness vary and may range from light-headedness and disorientation to balance problems and vertigo (Maskell et al., 2006). Recent work (Kleffelgaard et al., 2017) revealed dizziness-related disability is associated with increased vertigo symptoms, balance deficits, and psychological distress.
Dizziness can have many different plausible explanations; however, our approach is to help alleviate musculoskeletal problems that may be the underlying cause. To explain this rationale, we need to look at the anatomy of the neck and back and how chronic muscle tightness could impact functions related to dizziness. Firstly, we know that muscle tightness can occur as a result of overstretching the muscle during a movement resulting in muscle fibres or tendons torn, which may lead to inflammation and loss of muscle function (Bhimani & Carney-Anderson, 2017). Furthermore, it has been shown that sudden impacts can place strain on individuals’ muscles and likely is proportional to the amount of force sustained (Hedenstierna, Halldin, & Siegmund, 2009). Therefore, if a person sustains a force that overloads or overstretches the muscles, that individual could experience muscle tightness.
The exact symptoms resulting from muscle tightness depends on which muscles or areas are affected. We argue in this paper that dizziness could be a by-product of muscle tightness surrounding the neck area. Dizziness can have a wide array of causes that include, but limited to inner ear problems, drop in blood pressure, anemia, etc. (e.g., Cheever et al., 2016; Giza et al., 2014; Reneker et al., 2018); however, one of the causes that we are interested in is a dysfunction of the upper cervical spine in the case of cervicogenic dizziness (Ryan & Cope, 1955; Marshall et al., 2015; Treleaven, 2017). Since we know that muscle tightness may affect muscle function and can lead to inflammation (Bhimani & Carney-Anderson, 2017), this can have serious consequences for any structure that is in the surrounding area. For example, the sternocleidomastoid (SCM) is a muscle that has many many nerves running below it (e.g., the vagus nerve, spinal accessory nerve, greater auricular nerve) and blood supplies going to and from the head (e.g., posterior jugular vein, carotid artery; Agur & Dalley, 2009). Furthermore, these nerves and blood supplies innervate or affect other muscles such as the levator veli palatini muscle (innervated by the vagus nerve) which runs deep to the SCM (Agur & Dalley, 2009). One of the functions of the levator veli palatini is to help proper maintenance of the Eustachian tube inside the ear, which may contribute to dizziness if not working properly (e.g., Williams et al., 2020). Therefore, having muscle tightness can have other unintended consequences if not treated properly.
One method used to alleviate muscle tightness is soft tissue release. Soft tissue release is when a therapist actively stretches muscles and surrounding structures under a low load (Barnes, 1990; Ajimsha, Al-Mudahka, & Al-Madzhar, 2015; McKenney, Elder, Elder, & Hutchins, 2013) and can involve the use of trigger point therapy to localized areas of tightness (Wilhite, Paloncy, Rawkowski, & Daniel, 2019). There are multiple variations of this technique with some wide-ranging benefits listed in the literature. For example, providing better mobility (e.g., Domaszewska, Pieńkowski, Janiak, Bukowska, & Laurentowska, 2019; Wilhite, Paloncy, Rawkowski, & Daniel, 2019), increasing range of motion (de Souza et al., 2019), and alleviating pain (e.g., Ajimsha, Al-Mudahka, & Al-Madzhar, 2015; Kameda & Tanimae, 2019; Wasserman, Copeland, Upp, & Abraham, 2019). There are certainly limitations to soft tissue release and trigger point therapy; however, the benefits of using such an approach coupled with endurance training to maintain normal muscle function are not well understood in caring for individuals with a mTBI. Specifically, we are interested in asking if alleviating muscle tightness can result in reducing some feelings of dizziness by having better muscle functioning and blood flow throughout the neck region as it has been argued that combining cervical spine physiotherapy with vestibular rehabilitation may facilitate recovery in individuals with persisting dizziness following mTBI (Schneider et al., 2014).
In order to ascertain a better understanding of how alleviating muscle tightness can impact dizziness in mTBI it is necessary to compare subjective reports before and after a therapy, treatment, or intervention. The emphasis of such treatment is on resolving symptoms and remaining symptom-free (McCrory et al., 2017). The current research examined dizziness in clients with mTBI before and after an intensive daily treatment program short stay (i.e., 2-4 weeks) at a physiotherapy clinic in Southwestern Ontario, which included soft tissue release, specialized electrotherapy (Myoworx® TM20) and exercise therapy. It was hypothesized that scores from the Dizziness Handicap Inventory (Jacobson & Newman, 1990) would decrease following completion of the treatment program. As age and sex are often found to be associated with worse post-concussion symptoms, including dizziness (Cancelliere et al., 2016; Tinetti et al., 2000), we expected to see age- and sex-related effects in our results. Lastly, we know that severity of in injury, time since injury is a factor that influences recovery (Chamelian & Feinstein, 2004; Maskell et al., 2006).
Methods
Participants and procedures.
An application for secondary use of data from a physiotherapy clinic in Southwestern Ontario was approved by the University Ethics Board. Upon receiving ethics clearance, clinical data from 103 patients were sent electronically by the data custodian via a secure, password-protected, file transfer service to the first author. All patients had signed an agreement with the clinic for data to be used for research purposes. Identifying features of the dataset were removed, except for sex, age, and identification number.
The current research included clients who were between the ages of 13 and 62 years (Table 1) at intake (Children & Youth ages 13-24; Adults ages 25-64) with no known major medical issues (e.g., stroke, Multiple Sclerosis, etc.). It is important to note that all clients had other treatment prior to intake at the clinic and this was not their first attempt at alleviating dizziness symptoms (however it should also be noted that treatment providers gave clinical impression of post concussion syndrome and dizziness being one of the symptoms). Clients underwent an intensive daily treatment program with a short stay duration (i.e., 2 to 4 weeks) for soft tissue release with exercise therapy and specialized electrotherapy (Myoworx® TM20) to treat post-concussion syndrome (experience symptoms for longer than three months at least). The 75 patients whose records included a complete Dizziness Handicap Inventory (DHI; Jacobson & Newman, 1990) at both timepoints (i.e., pre- and post-treatment) were included in the present study. To mitigate the conflict of interest, the first author, who has no association with the clinic, was responsible for data analysis and interpretation of results; however, to ensure accuracy in the description of the therapy, the manuscript was prepared in collaboration with a clinic employees.
Table 1. Participant demographics
| N | Age at intake | Number of Concussions | Days Since Concussion | Days from Injury to 1st Tx | Days of in Tx Program | Tx to Date | |
| Total | 75 | 27.11 (10.17) | 2.69(2.00) | 763.71 (590.92) | 752.52 (590.45) | 11.19 (3.01) | 8.35 (2.42) |
| Male | 36 | 27.08 (8.11) | 2.75(1.96) | 729.14 (557.27) | 718.83 (557.27) | 10.31 (2.65) | 8.17 (1.96) |
| Female | 39 | 27.13 (11.85) | 2.64(2.03) | 795.62 (625.92) | 783.52 (625.16) | 12.00 (3.12) | 8.51 (2.79) |
| Children & Youth | 39 | 19.74 (3.32) | 2.67(2.11) | 738.90 (634.55) | 727.41 (633.54) | 11.49 (3.38) | 8.33 (2.64) |
| Male | 18 | 20.94(2.29) | 2.61 (1.61) | 633.67 (510.86) | 623.00 (510.87) | 10.67 (2.79) | 8.11 (2.19) |
| Female | 21 | 18.71(3.76) | 2.71 (2.49) | 829.10 (724.37) | 816.90 (722.88) | 12.19 (3.74) | 8.52 (3.01 |
| Adults | 36 | 35.08 (8.98) | 2.72 (1.88) | 790.58 (547.43) | 779.72 (547.43) | 10.86 (2.55) | 8.36 (2.19) |
| Male | 18 | 33.2 (7.12) | 2.89(2.30) | 824.61 (599.25) | 814.67 (599.10) | 9.94 (2.53) | 8.22 (1.77) |
| Female | 18 | 36.94 (10.39) | 2.56 (1.38) | 756.56 (505.40) | 744.78 (505.83) | 11.78 (2.29) | 8.50 (2.60) |
Myoworx® TM20 Soft tissue research with exercise therapy.
During the intake process the research aspect and rehabilitation approach were explained to all the clients. Each client was scheduled to receive treatment daily over a few weeks (2-3) as typically most clients analyzed consisted of travellers from outside of Ontario, and thus stayed in a nearby location where they could attend the clinic everyday. When visiting the clinic, most clients did not receive any other form of health care (not enforced) due to travelling from different parts of the globe.
Prior to commencing any treatment, all clients received a comprehensive assessment that was performed by a registered physiotherapist. The comprehensive assessment consisted of questionnaires and physical assessments based on their reported symptomology during the intake process. This assessment was used to guides the rehabilitation approach needed for the symptoms reported. Following the initial assessment, the clients were connected to a TM20 device. The TM20 device is designed to provide electrical stimulation for the purpose of muscle relaxation, similar to inferential current therapy with some proprietary modifications. It is unfeasible for this paper to report the intensity level of the TM20 as the levels are set differently for all the clients and on different days, as it is based on the clients’ self-report of how the vibrations feel on their muscles. However, the setting of the TM20 is based on operating below sub-threshold levels for muscle contractions; thereby no muscle contractions are invoked during this treatment (we reiterate that the purpose is to try to help muscle relaxation).
Following a 30-minute treatment with the TM20, the clients worked with a Registered Kinesiologist under with the guidance of the physiotherapist (i.e., delegated care). Although the rehabilitation approach was individualized for each client’s needs, the general approach was to relieve the root cause of the symptoms. This approach centers around the notion that the muscles on the back and the neck are affected (e.g., increased myofascial tension) following a concussion and associated symptoms (e.g., whiplash) and therefore require a person to re-learn how to relax and strengthen these muscles using targeted stretching and endurance protocols. Therefore, myofascial release techniques and muscle strengthening/endurance are used to help relieve symptoms.
The Dizziness Handicap Inventory (DHI)
The DHI (Jacobson & Newman, 1990) measures an individuals’ self-perceived level of handicap associated with dizziness; it is the most widely used self-report measure (Mutlu & Serbetcioglu, 2013). The questionnaire includes 25 items, with three response levels (i.e., 0 = no, 2 = sometimes, 4 = always), grouped into functional, emotional, and physical subscales. The sum of total possible scores range from 0 to 100, where higher scores are indicative of greater self-perceived level of handicap. According to Whitney et al. (2014), scores between 0 and 30 reflect mild handicap, 31 to 61 as moderate, and severe scores ranging from 61 to 100. Scores are associated with levels of functional balance impairment (Whitney et al., 2014).
Data Analysis
Subscale (i.e., functional, emotional, and physical) scores and a total score were computed from the DHI. Scores were submitted to mixed Analysis of Variance (ANOVA) tests, with measure (i.e., pre x post) as the within subject measure, and age group (i.e., children/youth x adult) and sex (male x female) as the between subjects’ measure. The number of mTBIs and days since last mTBI were entered as covariates. Subsequent univariate ANOVAs were performed to focus exclusively on the effect of measure (i.e., pre x post).
Results
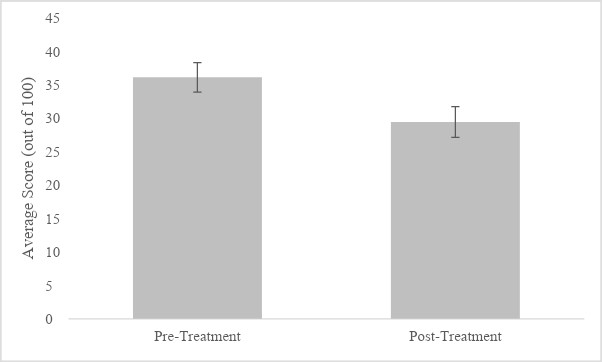
When controlling for the number of mTBIs and days since the last mTBI, analysis of total scores revealed no statistically significant change in total DHI score (F (1, 69) = 3.282, p = .074, η2p = .045). However, as evidenced in pairwise comparisons (MDifference = -7.91, 95% CI [-4.25, -11.58]), total DHI scores decreased from the moderate range (M = 36.83, SD = 19.41) to the mild range (M = 28.96, SD = 20.74) after treatment, reflecting a clinically significant change (Figure 1). No other significant effects emerged (p > .05)
No differences emerged between the age groups (F (1, 69) = 3.325, p = .073, η2p = .046) or as a function of sex (F (1, 69) = 2.047, p = .157, η2p = .029). Likewise, the co-variates number of mTBIs (F (1, 69) = 2.134, p = .149, η2p = .030) and days since last mTBI (F (1, 69) = 0.885, p = .350, η2p = .013) were not significant predictors. None of the interactions were significant (p > .05).
It is important to note that, despite lacking statistically significance, total DHI scores were initially in the moderate range (M = 36.83, SD = 19.41), decreasing to the mild range (M = 28.96, SD = 20.74) after treatment. Data were thus re-analyzed without the between subject factors and covariates (as previously stated, time since concussion and severity can influence recovery; Chamelian & Feinstein, 2004; Maskell et al., 2006); results revealed DHI total scores decreased over time (F (1, 74) = 18.680, p < .001, η2p = .202; MDifference = -7.87, 95% CI [-4.24, -11.49]; Figure 1).
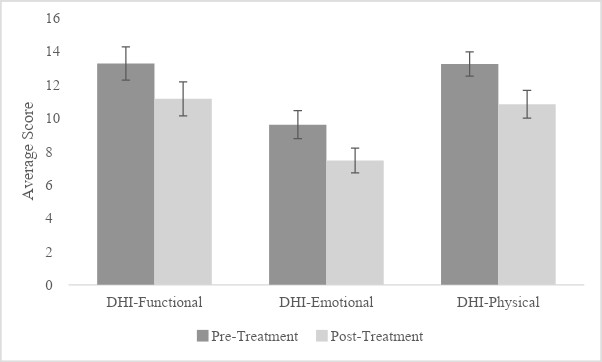
DHI subscale scores were also assessed (Figure 2). When considering between-subjects factors and covariates, controlling for the number of mTBIs and days since last mTBI, analysis revealed no statistically significant change in the functional subscale (F (1, 69) = 2.174, p = .145, η2p = .031). Analysis of subscales revealed no significant effects or interactions in functional. However, as evidenced in pairwise comparisons, a clinically relevant decrease in scores was observed (MDifference = -2.47, 95% CI [-0.88, -4.06]). Similarly, there was no statistically significant difference in emotional domain scores (F (1, 69) = 0.948, p = .334, η2p = .014); however, as evidenced in pairwise comparisons, changes were clinically important ((MDifference = -2.67, 95% CI [-1.240, -4.10]). There was a significant decrease in physical subscale as scores were significantly lower after treatment (M = 10.64, SD = 7.34) compared to the initial assessment (M = 13.39, SD = 6.38; F (1, 69) = 5.234, p = .025, η2p = .071; MDifference = -2.77, 95% CI [-1.50, -4.04]). Female participants had significantly greater physical sub-scores (M = 13.723, SE = 0.975) compared to male participants (M = 10.292, SE = 1.012; F (1, 69) = 5.943, p = .017, η2p = .079; MDifference = -3.43, 95% CI [-0.62, -6.24]). No other significant effects emerged (p > .05)
Subsequent univariate ANOVAs focusing exclusively on the change in score over time revealed a decrease in functional (F (1, 74) = 9.976, p = .002, η2p = .119; MDifference = -2.45, 95% CI [-0.91, -4.00]), emotional (F (1, 74) = 14.286, p < .001, η2p = .162; MDifference = -2.67, 95% CI [-1.26, -4.07]), and physical (F (1, 74) = 18.314, p < .001, η2p = .198; MDifference = -2.75, 95% CI [-1.47, -4.03]) subscales after treatment (Figure 2).
Discussion
The current research assessed dizziness in clients with mTBI before and after a short stay (i.e., 2-4 weeks) at a physiotherapy clinic with Myoworx® TM20 soft tissue release and exercise therapy. When controlling for number of concussions and days since concussion, there was no statistically significant change in total DHI scores. This is not surprising as it is well known that time since concussion and severity can influence recovery (Chamelian & Feinstein, 2004; Maskell et al., 2006). Therefore, our data supports that the longer a person waits for treatment, the less impact this type of rehabilitation program will have on dizziness. Even so, as health practitioners it is unethical to withhold treatment from clients and it is important to understand if such treatment can result in positive changes. Therefore, it also important to note that scores were observed to decrease from the moderate to mild range after treatment. It can be argued that, suggesting clinically significant changes or clinically meaningful differences were observed. While it important to note that there are currently no standards for computing such effect (Page, 2014), confidence intervals are generally accepted to provide information about clinical significance (du Prel et al., 2009; Page, 2014). Analyses were subsequently performed without covariates or between subjects’ factors; results were consistent with our hypothesis, revealing a decrease in total scores after treatment.
When DHI scores were separated into subscales, statistically significant changes were not observed in functional or emotional domains when controlling for number of concussions and days since concussion. As evidence in pairwise comparisons, clinically relevant changes were identified in functional and emotional domains. When analyses were repeated without covariates or between subjects, results were consistent with our hypothesis (i.e., decrease after treatment). Previous research has noted that physical and functional scores are most often greater than emotional scores (Loughran et al., 2006; Ten Voorde et al., 2012). For example, over fifty percent of patients in assessed by Van Voorde et al., (2012) reported low scores (i.e., mild impairment) on the emotional subscale; however, physical and functional scores were more often in the moderate or severe range. Likewise, Lin et al. (2002) found functional complaints most prevalent, but also found emotional subscales were lowest. Although improvement was indeed displayed in all subscale scores, it can be suggested that when controlling for number of concussions and days since concussion, perceived physical deficits emerged as the greatest effect on improvement in the current research. When analyses were repeated without covariates or between subjects, results were consistent with our hypothesis (i.e., decrease after treatment).
The use of myofascial release techniques and muscle strengthening/endurance have little to no adverse events compared to other modalities (e.g., drugs) for decreasing muscle tightness (citation). Our data supports that at the very least it may not reduce dizziness, but at the very best, it may lower dizziness scores for clients. Therefore, we recommend that understanding the anatomy of the neck/shoulder/back regions are imperative in treatment of mTBI from a rehabilitation point of view. A base understanding of physiology, neuroscience, and anatomy can help shape hypothesis and guide treatment of symptomology. This type of treatment is not new as Wrisley et al., (2000) describe a 49-year-old with a diagnosis of cervicogenic dizziness (i.e., “a non-specific sensation of altered orientation in space and disequilibrium originating from abnormal afferent activity from the neck;” Forman & Cass, 1996 c.f. Wrisley et al., 2000, p. 755) after a motor vehicle accident. Similar to the current treatment, physical therapy included “soft tissue massage, mobilization, and deep massage to her cervical musculature with emphasis on massaging the trigger points in the sternocleidomastoid muscle.” (p. 763). At home, she engaged range of motion exercises, performed balance activities, began a walking program, and was provided a transcutaneous electrical nerve stimulation (TENS) unit to alleviate pain and spasms in cervical muscles. After 3 weeks, pain levels decreased and the patient only reported one episode of dizziness (Wrisley et al., 2000). Furthermore, a systematic review (Reid & Rivett, 2005) discussed self-report improvement after cervical manipulations and exercises. Likewise, Schneider et al. (2014) identified that patients who engaged in a combination of vestibular and cervical treatment were 3.91 times more likely to return to activities daily life in less than 8 weeks compared to those who did not have therapy. Results from the current study provide additional evidence that manual therapy combined with exercise is beneficial to decrease dizziness in individuals with mTBI, particularly physical symptoms.
Our secondary hypothesis was related to the influence of age and sex, as both factors are typically related to more debilitating post-concussion symptoms, including dizziness (Cancelliere et al., 2016; Tinetti et al., 2000). Analysis revealed the physical scores of female participants were significantly greater than those of male participants; however, the effect of age was not significant. It has been argued that women are typically more affected by dizziness, and thus seek medical advice and assistance more frequently than men (Mutlu & Serbetcioglu, 2013; Loughran et al., 2006; Ten Voorde et al., 2012). Related research has noted higher DHI scores in female compared to males (Piker et al., 2008; Ten Voorde et al., 2012) as well.
With respect to age-related effects, relatively lower levels of impairment have been reported observed in very young patients (i.e., younger than 20 years), and relatively higher levels in very old (i.e., older than 80 years; Ten Voorde et al., 2012). Whitney et al. (2002) revealed the physical subscale score was worse in older adults ages 60 to 80. Participants in the current study ranged in from 13 to 62 years, with an average age of about 27, therefore perhaps there is a window where rehabilitation becomes less impactful. Furthermore, DHI scores at intake were in the moderate range (M = 36.83, SD = 19.41) and it could suggest that the moderate scores were reflective of the majority of participants being in the “middle” age range. Ten Voorde et al., (2012) reported a S-shaped relationship between age and total DHI score in 9- to 97-year-olds, albeit patients were suffering from dizziness as a result of varying diagnoses. Whitney et al. (2002) revealed the physical subscale score was worse in older adults ages 60 to 80. That said, others have not identified an no association between age and DHI (Loughran et al., 2006), with our evidence supporting this point of view.
Overall, findings from the current literature offer additional support for the notion that manual therapy combined with exercise is beneficial to decrease dizziness in individuals with mTBI. While significant differences after treatment were only identified in physical symptoms, confidence intervals were used to infer clinically meaning effects. Female participants had greater physical scores overall; yet, age-related effects were not observed. Together, findings are of practical relevance to the many clients who have and will continue to frequent the clinic for Myoworx TM20 and exercise therapy to help alleviate symptoms of mTBI.
Conflict of Interest
Some of the authors of this paper were or are currently employed in the clinic where the research data was collected. However, the leading author was asked to be involved with the project as a means to mitigate the conflict of interest that would exist if MMTR were to independently analyze and publish their clinical data.

